
Krebsforschungs-Lösungen
Verständnis zellulärer Signalwege und Interaktionen in der Krebsforschung mit 3D-Imaging
Krebstherapeutisches Screening mit 3D-Imaging von Sphäroiden, Organoiden und Organ-on-a-Chip-Systemen
An Krebs sind Veränderungen beteiligt, die es Zellen ermöglichen, sich ohne Rücksicht auf normale Einschränkungen zu teilen, angrenzende Gewebe zu befallen und zu zerstören und letztendlich Metastasen an entfernten Körperstellen zu bilden. Krebsforscher benötigen Werkzeuge, die es ihnen ermöglichen, die komplexen und oftmals schlecht verstandenen Interaktionen zwischen Krebszellen und deren Umgebung einfacher zu untersuchen und therapeutische Interventionspunkte zu identifizieren.
Lernen Sie unser automatisiertes Zellkultursystem, unsere High-Content-Imaging-Systeme und unsere Analysesoftware-Lösungen kennen. Sie vereinfachen die Krebsforschung, wenn biologisch relevante 3D-Zellmodelle wie Sphäroide, Organoide und Organ-on-a-Chip-Systeme angewandt werden, um die In-vivo-Umgebung eines Tumors oder Organs zu simulieren.

Aus Patienten stammende kolorektale Krebsorganoide. Blau: Zellkerne, grün: Aktin, rot: Mitochondrien.
Vorteile der 3D-Imaging-Technologie für Krebs-Sphäroide
Krebs-Sphäroide imitieren das Verhalten von Tumoren wesentlich wirksamer als Standard-2D-Kulturen. Solche 3D-Sphäroid-Modelle werden erfolgreich für das Screening von Umgebungen eingesetzt, um potentielle Krebstherapeutika zu identifizieren. Diese Kultursysteme können für multiparametrische Analysen genutzt werden, um verschiedene biologische Messungen zu quantifizieren und dadurch die Entwicklung von Wirkstoffen gegen Krebs zu beschleunigen.
Zu den wichtigsten Vorteilen zählen:
- Die Entwicklung des 3D-High-Content-Imaging stellt einen wichtigen Schritt dar, um relevantere und präzisere Tests zu ermöglichen
- 3D-Kultursysteme können schnell einheitliche Krebs-Sphäroide aus menschlichen Krebszellen erzeugen, die in Hochdurchsatz-Formaten eingesetzt werden können, um die Entwicklung von Wirkstoffen gegen Krebs zu beschleunigen
- Die Forschung hat mithilfe der konfokalen 3D-Bildanalyse von Krebszellen die multiparametrische Beschreibung vieler biologischer Ergebnisse ermöglicht
Arbeitsablauf zur Analyse von 3D-Krebsphäroiden in einer Hochdurchsatz-Screening-Umgebung
Sphäroide können in 96- oder 384-Well-Platten gezüchtet, mit Verbindungen behandelt und mit Farbstoffen gefärbt werden, die aktive zelluläre Prozesse und Signalwege sichtbar machen. In manchen Fällen können Sphäroide ohne vorheriges Waschen aufgenommen werden, und sie können, falls erwünscht, auch fixiert werden.
Vereinfachen Sie Ihre Arbeitsabläufe in der Onkologie – mit einem breiten Spektrum an Imaging-, zellulären Screening- und Mikroplatten-Reader-Systemen.
Der Arbeitsablauf illustriert einen vereinfachten Prozess zur Analyse von Sphäroiden und beleuchtet Systeme mit dem Ziel, Ihnen bei der Optimierung Ihrer Forschung zu helfen und Ihren Durchsatz zu erhöhen.
Sphäroide kultivieren – Krebszellen können direkt in einer Platte mit äußerst geringer Bindung (ULA, ultra-low attachment), in Rundbodenplatten oder anderen Kulturplatten kultiviert werden, um die typische Morphologie eines Sphäroids zu entwickeln. Andere Kultivierungsplatten ermöglichen es, mehrere Sphäroide in einem einzigen Well wachsen zu lassen.
Mit Verbindungen behandeln – nachdem die Sphäroide ausgebildet sind, werden Verbindungen in den gewünschten Konzentrationen zu den Wells hinzugefügt und für einen Tag bis mehrere Tage inkubiert, je nachdem, welcher Mechanismus untersucht wird.
Auf Marker färben – nachdem die Behandlung mit Verbindungen abgeschlossen ist, werden die Farbstoffe direkt zum Medium hinzugegeben. Um eine Störung der Sphäroide zu vermeiden, können Färbungen angewendet werden, die keinen Waschschritt erfordern. Sphäroide können, wenn notwendig, jedoch auch vorsichtig gewaschen werden, sogar automatisiert.
Sphäroid-Abbildungen aufnehmen – mittels spezialisierter Imaging-Ausrüstung können Bilder vom Inneren des Sphäroid-Körpers einzeln oder als ein Z-Stapel (mehrere Bilder in verschiedenen Tiefen) aufgenommen werden.
Krebszellen analysieren – nutzen Sie eine Software für zelluläre Bildanalyse, um die Zellbilder quantitativ auszuwerten, um die Expression verschiedener Marker zu überwachen und um biologische Messungen zu quantifizieren.
Konfokales High-Throughput-Imaging von Sphäroiden für das Screening von Wirkstoffen gegen Krebs
In den letzten Jahren wurde ein wesentlicher Fortschritt in der Entwicklung von In-vitro-Aggregaten aus Tumorzellen erzielt, die als Modelle für die In-vivo-Gewebeumgebung dienen. Wenn diese Aggregate in ein Well einer Platte mit Rundboden und äußerst geringer Bindung ausgesät werden, bilden sie jeweils ein separates Sphäroid. Von Sphäroiden wird angenommen, dass sie das Verhalten von Tumoren wirksamer nachahmen als reguläre zweidimensionale (2D)-Kulturen. Der Grund ist, dass sie, wie Tumore, sowohl an der Oberfläche exponierte als auch tief verborgene Zellen, proliferierende und nicht-proliferierende Zellen, ein sauerstoffarmes Zentrum und eine gut mit Sauerstoff angereicherte äußere Zellschicht enthalten. Solche 3D-Sphäroid-Modelle werden erfolgreich für das Screening von Umgebungen eingesetzt, um potentielle Krebstherapeutika zu identifizieren.
Während die Entwicklung robuster Sphäroid-Assays einige Herausforderungen mit sich bringt, ist der Einsatz des automatisierten High-Throughput, High-Content-Imaging ein wesentlicher Schritt, um relevantere Tests auf chemotherapeutische Wirkstoffkandidaten zu erleichtern.
- Nehmen Sie ein komplettes Sphäroid bei einer 20-fachen Vergrößerung in einem Gesichtsfeld auf
- Screenen Sie biologisch relevante 3D-Sphäroide im 96- oder 384-Well-Format
- Nutzen Sie konfokales Imaging, um zelluläre Antworten präzise zu detektieren
- Sparen Sie Speicherplatz, indem Sie lediglich 2D-Rekonstruktionen von Z-Ebenen-Abbildungen speichern
Abbildung 1. Screenen Sie 3D-Sphäroide schnell in Mikroplatten
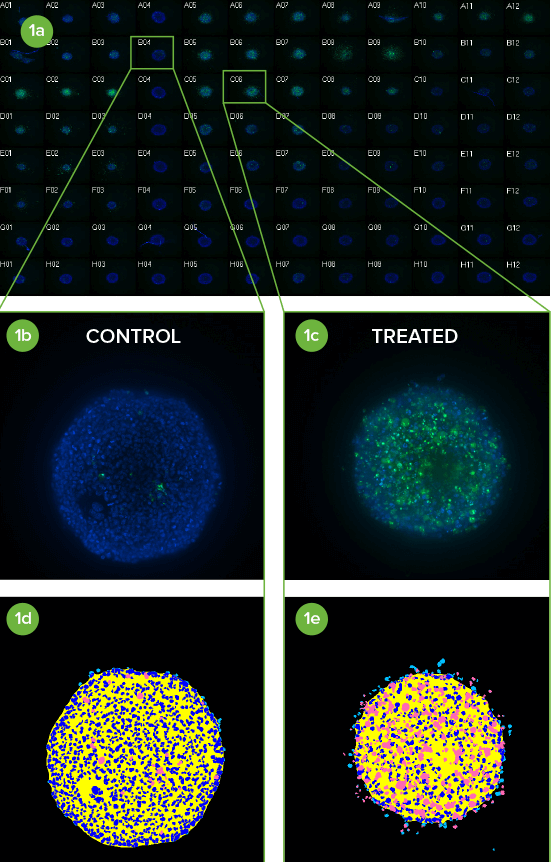
(1a) Zusammenstellung aus Miniaturansichten von HCT116-Sphäroiden in einer 96-Well-Platte, die mit Verbindungen behandelt und mit einem 10x Plan Fluor-Objektiv aufgenommen wurden. Mit Hoechst gefärbte Zellkerne (blau) sind mit dem Apoptose-Marker CellEvent Caspase 3/7 (grün) überlagert.
(1b) Unbehandelte Kontrollen sind in Spalte 4 aufgezeigt und (1c) eine Caspase 3/7-Antwort ist in den Spalten 5–7 ersichtlich, in denen Paclitaxel ausgehend von 1 µM in Reihe A 1:3 seriell verdünnt wurde (in waagerechten 3er-Replikaten).
(1d, 1e) Elf Z-Ebenen wurden in einem 2D-Bild mit maximaler Projektion kombiniert und mithilfe eines einfachen benutzerdefinierten Moduls analysiert. Gezeigt sind Rohabbildungen, die mithilfe der entsprechenden Segmentierungsmasken einen niedrigen und hohen Grad an Apoptose anzeigen (königsblau = Zellkerne, pink = apoptotische Zellen).
Anwendungen und Assays
Molecular Devices, ein Branchenführer für Lösungen im Bereich der komplexen Biologie, stellt ein breites Spektrum an Werkzeugen zur Unterstützung der Life Science-Forschung, Wirkstoffforschung und des High-Throughput-Screenings zur Verfügung. Unsere KI-gesteuerte automatisierte Zellkultur und unsere High-Content-Imaging-Systeme können den Erfolg Ihrer bioanalytischen Krebsforschung vorantreiben. Wir bieten darüber hinaus etliche Konfigurationen für Ihre Multi-Mode Mikroplatten-Reader sowie eine Reihe von einfach zu bedienenden Microarray-Scannern.
Erfahren Sie mehr darüber, wie unsere Technologie Ihre Forschung an Krebs-Therapeutika unterstützen kann.