
Application Note
Improving the robustness of Cell Painting with a near-infrared label and advanced image and data analytics
- Improve the sensitivity of the Cell Painting assay using a near-infrared label.
- Get robust image segmentation using IN Carta’s deep learning SINAP module
- Easy to use cloud-based data analytics software with StratoMineR.
Introduction
Image-based phenotypic profiling approaches such as the widely used Cell Painting assay use high-content imaging along with multiparametric readouts to study biological, genetic, and chemical perturbations in cells. This increasingly popular method is being used in applications ranging from drug discovery programs to genomic screening studies.
In the standard Cell Painting assay, a variety of cellular organelles and structures (nuclei, cytoplasm, mitochondria, Golgi, ER, cytoskeleton, cytoRNA, and nucleoli) are captured in five imaging channels. This broad staining of the cell enables multiple morphological perturbations to be monitored and quantified at single-cell resolution. Limitations of the published Cell Painting assay include the narrow availability of suitable dyes and the ability of imaging systems to give adequate spectral separation. Specifically, Golgi and actin are imaged using the same channel, which poses a challenge when separating the two structures during image analysis. This limitation may interfere with hit selections or mask the effects of some compounds— especially those with a mechanism of action (MOA) that affects the Golgi (biosynthetic pathways) or cytoskeletal network.
Here, we sought to improve the assay by taking advantage of the ImageXpress® Confocal HT.ai High-Content Imaging System which is equipped with a NIR light source. We swapped out Alexa Fluor 568 Phalloidin for Alexa Fluor 750 Phalloidin which allows for the cytoskeleton to be distinctly separated from the Golgi compartment.
Due to the complexity of this multiplexed assay, the sheer volume of data generated from analysis of these images requires additional analytical tools to help mine and interpret the data. For manageable image and data analyses, we used IN Carta® Image Analysis Software for feature extraction, followed by HC StratoMineR™ for data mining and analysis. IN Carta includes a deep-learning function that allows for more robust feature segmentation. The Cell Painting assay thus benefits from improved nuclei detection when used with the deep learning SINAP module. Furthermore, organelles such as mitochondria, ER, Golgi, and the cytoskeleton can be further segmented using deep-learning functionality if required. Measurements that encompass fluorescent intensity, spatial numbers, texture, organelle distribution, and colocation measures may be extracted from the analyzed images. The data is then uploaded into HC StratoMineR, an intuitive cloud-based data analytics platform for further data analysis. To determine if our approach demonstrates any advantages over standard Cell Painting protocols, we compared the phenotypic distance scores between images acquired with five channels and those acquired with six channels. We found up to 49% increase in distance score for cells treated with a subset of compounds. These results show that using separate imaging channels for the Golgi and cytoskeleton increases the sensitivity of the Cell Painting assay and more robustly represents diverse cellular phenotypic profiles.
Methods
- U2OS cells (ATCC) were seeded at 2000 cells per well.
- Eleven compounds were tested in quadruplicate wells in a seven point 1:3 dilution series along with suitable controls. Compounds used: Ca-074-Me, CCCP, chloroquine, cytochalasin D, etoposide, latrunculin B, rapamycin, rotenone, staurosporine, paclitaxel and tetrandrine.
- Cells were stained using the protocol by Bray et al1. For the modified protocol, phalloidin/Alexa Fluor Plus 750 (Thermo Fisher) was used instead of phalloidin/Alexa Fluor 568.
- Image acquisition was performed on either the ImageXpress Confocal HT.ai (laser based) or ImageXpress Micro Confocal (LED based) high-content imaging system (Molecular Devices) using a 20X Plan Apo objective. The filters used are indicated in Table 1.
- IN Carta Image Analysis Software was used for image analysis. Measurements that were selected include parameters related to intensity, texture, shape, spatial relationship, and co-localization scores.
- Cell-level data was uploaded into StratoMineR™ (https://corelifeanalytics.com/applications/cell-painting/, Core Life Analytics) for further data analysis. Briefly, quality control, plate normalization, data transformation, and feature standardization was applied. Principal component analysis (PCA) was used to reduce the dimensionality of the data set. Further downstream analyses such as hit selection and cluster analysis were performed based on the principal components and the phenotypic distance scores.
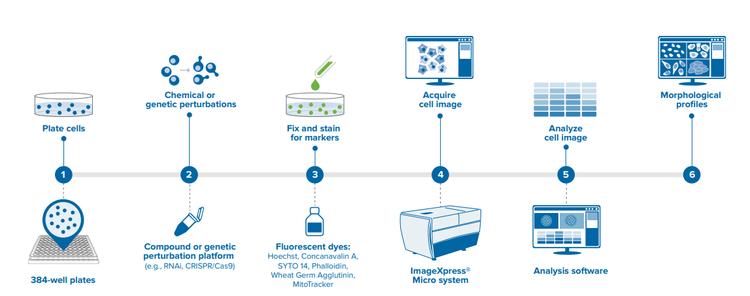
Overview of the workflow for image-based profiling. Details for each step listed above.
Results
Image acquisition
The Cell Painting assay uses six fluorescent markers to simultaneously label the cell, and the various cellular structures are then imaged. The Golgi apparatus (AGP channel) and cytoskeleton are both imaged using the same filter set (Table 1, Figure 1). Resolution of these subcellular structures is typically carried out in the image analysis step. In our model assay, U2OS cells were treated with 11 compounds for 24 hours and then processed according to a previously published protocol.1, 2 Cells were then imaged using five imaging channels (Figure 1).
To separate the Golgi structures from the cytoskeleton, we modified the assay by swapping out Alexa Fluor 568 Phalloidin for Alexa Fluor 750 Phalloidin and image the cells using the ImageXpress Confocal HT.ai High-Content Imaging System equipped with a NIR light source (Figure 2A) (hereafter referred to as the modified assay). With this approach, the Golgi structures can be clearly seen in a perinuclear distribution in untreated cells. This distribution is not easily distinguished when both WGA (for Golgi) and phalloidin (for actin) stain were imaged in the same channel (Figure 1). The difference is more apparent with compound (e.g. Tetrandrine) treated cells (Figure 2B)
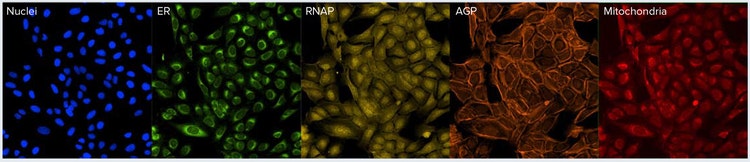
Figure 1. Cell Painting assay. Examples of cells stained with dyes for the cell painting assay using previously published protocols. Note that Golgi and actin structures are acquired in the same channel (AGP). Actin staining is represented by the filamentous structures. However, the Golgi is not easily distinguished by eye.
516/549 (DNA)
521/547 (RNA)
Table 1. Stains used in the Cell Painting assay and filter sets used for detection of cellular compartments
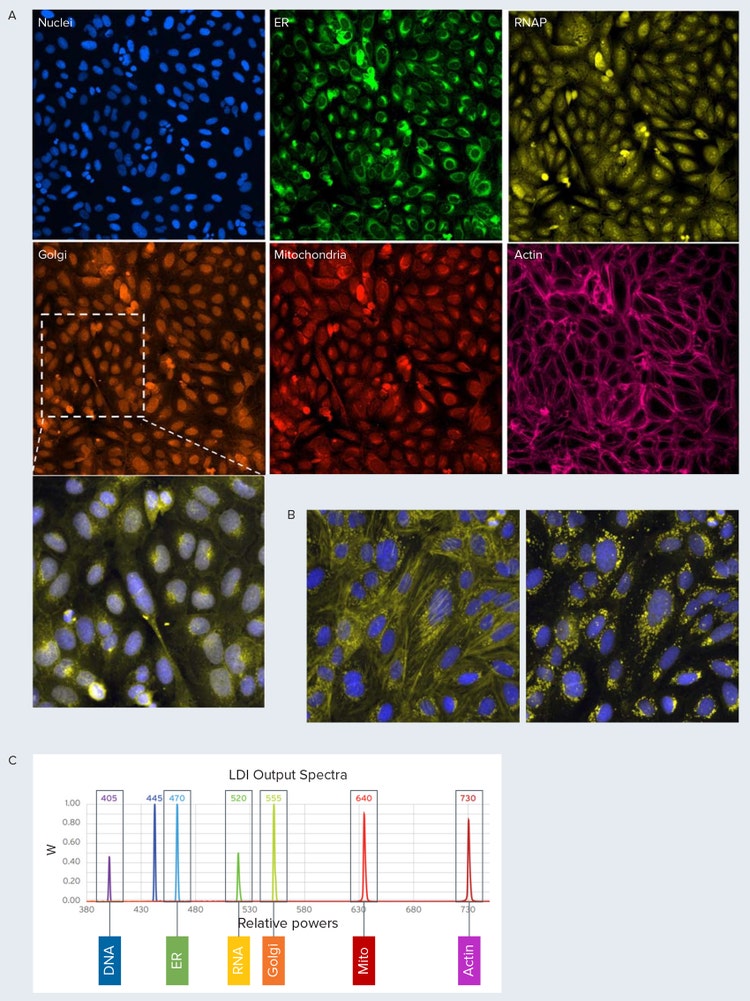
Figure 2. Cell painting assay modified Modified protocol using far-red light source to improve spectral separation in the Cell Painting assay. A) Phalloidin Alexa Fluor 750 was used to label actin in the modified protocol. Example images from a control well is shown. Inset (white box) is zoomed in and shown as a composite of the nuclei (blue) and Golgi (yellow) channel. Golgi punctae are clearly seen distributed in a mostly perinuclear organization. B) Images of cells treated with tetrandrine. On the left is a reconstructed image of the actin and Golgi channel merged (yellow). Right shows the image with only Golgi (yellow) and nuclei (blue). The effects of tetrandrine on Golgi distribution is clearer with the modified protocol (right). C) Laser light source and their relative powers shown. Detection of cellular compartment with their respective light source indicated
Feature Extraction and data analysis
To quantify the differences between cells stained with the standard cell paint dye set vs. those stained with the modified dye set, images were analyzed in the IN Carta Image Analysis Software (Figure 3). Extracted measurements were uploaded into HC StratoMineR for further data analysis. Principal component analysis (PCA) was carried out on the data extracted from the standard Cell Painting assay vs. the modified assay. Interestingly, measurements from the AGP channel make up most of the feature components in PCA 1 from 5-channel images. However, measurements from the actin channel make up most of the dominant features in PCA 1 from the modified 6-channel images (Figure 4). This suggests that the modified assay can improve resolution of phenotypic profile between the actin and Golgi components which is useful in understanding MOA.
We also compared the phenotypic profiles between the standard and modified Cell Painting assay based on their distance scores. This score represents the phenotypic distance a sample is from the negative control which can be used for hit selection in screening assays. We found an increase in the distance score for compounds known to affect the Golgi pathway (Table 2). These results show that using separate imaging channels for the Golgi and cytoskeleton increases the sensitivity of the Cell Painting assay and more robustly represents the cellular phenotypic profiles.
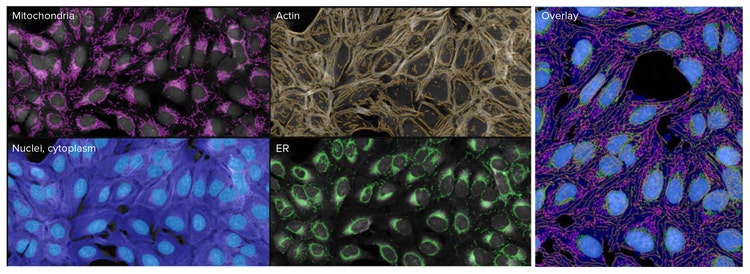
Figure 3. Feature extraction was done in the IN Carta software to segment the various cellular structures. Here, the built-in nuclei model was used to achieve robust segmentation of nuclei across all treatments. For images acquired in six channels, 487 features were extracted per cell. Shown here are example images with feature mask overlay in IN Carta software.
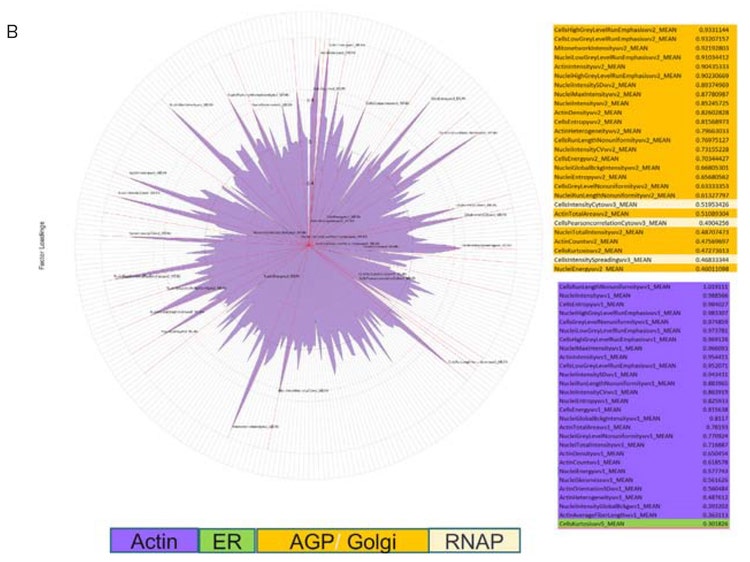
Figure 4. Using HC StratoMineR for data analysis. A) HC StratoMineR is a web-based platform which guides users through a typical workflow in analysis of high-content multi-parametric data. B) PCA (generalized weighted least squares) was used to reduce the data to 15 components. Feature contributions to PCA1 is shown as a polar plot. Features color coded according to the cellular compartment they represent. PCA1 features from 5 channel (top) and 6 channel (bottom) Cell Painting acquisition. Note the differences in the feature contributions.
Table 2. Change in the distance score between the data set acquired using 5-channels and 6-channels for Cell Painting.
Conclusions
- Our results demonstrate improvement in sensitivity to the Cell Painting assay with the use of a near-infrared laser. In this case it allowed for the development of an assay that is more sensitive in the separation of Golgi phenotypes.
- The extra channel will also allow for the extension of the standard Cell Painting assay through the addition of project-specific biomarkers.
References
- Bray MA et.al., Nat Protoc. 2016 Sep;11(9):1757–74
- Gustafsdottir SM et.al., PLoS One. 2013 Dec 2;8(12):e80999.
- Omta WA, et.al., Assay Drug Dev Technol. 2016 Oct;14(8):439–452